Home
Engineering the Future of Precision Medicine
VJLabs at the Terasaki Institute for Biomedical Innovation engineers next-generation organ-on-a-chip platforms and microphysiological systems that recapitulate human organ physiology and disease.
Founded in 2020 by Dr. Vadim Jucaud, a mentee of the late Dr. Paul I. Terasaki, the lab brings together immunology, microfluidics, tissue engineering, biomaterials, and biosensing to build in vitro models that accelerate drug discovery, reduce reliance on animal testing, and advance personalized medicine.
At the heart of VJLabs is our proprietary vascularized Tissueoid technology, which integrates perfusable microvascular networks with three-dimensional tissue constructs to create organ models with unprecedented physiological relevance. This platform serves as the foundation for five interconnected research programs spanning organ-on-a-chip engineering, transplant immunology, cancer biology, biosensing, and biomaterials.
Dr. Vadim Jucaud

Assistant Professor, Terasaki Institute for Biomedical Innovation
Dr. Vadim Jucaud is an Assistant Professor at the Terasaki Institute for Biomedical Innovation and the founder of VJLabs. He holds a Ph.D. in Immunology and Microbiology with specialization in transplant immunology and Human Leukocyte Antigen (HLA) immunobiology.
Dr. Jucaud began his research career in 2010 under the mentorship of Professor Paul I. Terasaki, working in the fields of histocompatibility and immunogenetics with a focus on HLA antibodies and the Humoral Theory of Transplantation. Over 12 years, he developed deep expertise in HLA immunogenetics, antibody cross-reactivity, immunogenic epitope characterization, and T and B cell immunomodulation.
In 2018, he received the American Transplant Congress Young Investigator Award for his work demonstrating the prevalence and impact of de novo donor-specific antibodies during a multicenter immunosuppression withdrawal trial in adult liver transplant recipients.
Motivated by this foundation and the multidisciplinary environment at TIBI, Dr. Jucaud established VJLabs in 2020 to pioneer immunocompetent organ-on-a-chip platforms at the intersection of HLA immunobiology, microfluidics, tissue engineering, biomaterials, and biosensing.
Five Interconnected Research Areas Driving Biomedical Innovation
Our research spans organ-on-a-chip engineering, immunology, biosensing, biomaterials, and cancer biology, all converging on the vascularized Tissueoid platform to create clinically relevant human tissue models.
Organ-on-a-Chip & Microphysiological Systems
Engineering the next generation of human-relevant tissue models
The central pillar of VJLabs. We engineer vascularized, perfusable organ models including liver, blood-brain barrier, lymph node, and glioblastoma that recapitulate human physiology and disease.
What: Vascularized Tissueoid platforms with perfusable microvascular networks, immune cell access, and integrated biosensors.
Why: Conventional models lack vasculature, immune competence, and real-time readouts, limiting their predictive value for drug discovery and regulatory applications.
How: Scalable thermoplastic fabrication, modular chip architectures, and accessible manufacturing designed for academic, pharmaceutical, and regulatory adoption.
Transplant Immunology & HLA Immunobiology
Decoding the immune response to improve transplant outcomes
We study how donor-specific HLA antibodies shape transplant outcomes by mapping antibody-antigen interactions, characterizing immunogenic epitopes, and developing computational models to predict immune responses.
What: HLA antibody characterization, epitope mapping, immunogenicity prediction, and computational donor-recipient matching using the E3 feature engineering software.
Why: Antibody-mediated rejection remains the leading cause of long-term graft loss. Predicting which mismatches trigger immune responses is critical for improving patient outcomes.
How: Our deep immunological expertise directly informs our organ-on-a-chip platforms, making them uniquely immunocompetent for modeling allograft rejection, tolerance pathways, and immunosuppressive strategies.
Cancer Biology & Immunotherapy
Personalized cancer models for better therapeutic decisions
We address critical gaps in cancer research through engineered tissue models for cancer vaccine screening, drug resistance studies, and embolic agent evaluation.
What: Lymph node-on-a-chip for cancer vaccine efficacy screening (including aging populations), glioblastoma-on-a-chip for pericyte-mediated drug resistance, and vascularized liver tumor model for embolic agent testing.
Why: Preclinical models fail to capture the complexity of human immune responses, especially in elderly patients who bear the highest cancer burden and are often overlooked in testing.
How: Our platforms recapitulate antigen presentation, T cell activation, tumor microenvironment mechanics, and age-dependent immune responses in controlled, human-relevant systems.
Biosensors & Real-Time Monitoring
Turning organ-on-a-chip platforms into continuously monitored living systems
We develop and integrate label-free sensing technologies directly into our organ-on-a-chip platforms for continuous, real-time monitoring without disrupting the biological system.
What: PC-TIR optical biosensors for antibody/toxicity monitoring, TEER electrodes for barrier integrity, electrochemical immunosensors, and contact lens biosensors for ocular biomarker detection.
Why: Traditional drug screening relies on endpoint assays that miss dynamic cellular responses, contributing to a >90% clinical trial failure rate.
How: Sensors are embedded directly into chip devices, enabling multiplexed readouts that generate richer mechanistic data for regulatory submissions and clinical decisions.
Biomaterials & Tissue Engineering
From advanced hydrogels to clinically translatable therapeutics
We engineer advanced biomaterials that enhance the physiological relevance of our tissue models and enable standalone clinical applications in wound healing and regenerative medicine.
What: Tunable hydrogel platforms (ionogels, peptide hydrogels), oxygen-generating microparticles, natural rubber latex therapeutics, and nanofibrous scaffolds for tissue repair.
Why: Biomaterials that faithfully replicate native tissue properties are essential for both accurate in vitro modeling and direct therapeutic applications.
How: We integrate biomaterials into microfluidic platforms for real-time evaluation and develop standalone dressings and scaffolds for clinical use, including a pilot trial for diabetic foot ulcers.
Five Core Technologies Powering VJLabs Research
Each technology addresses a specific barrier in biomedical research. Together, they form an integrated ecosystem for building clinically relevant human tissue models.
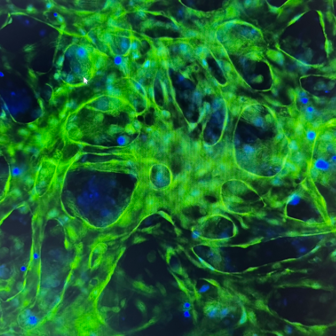
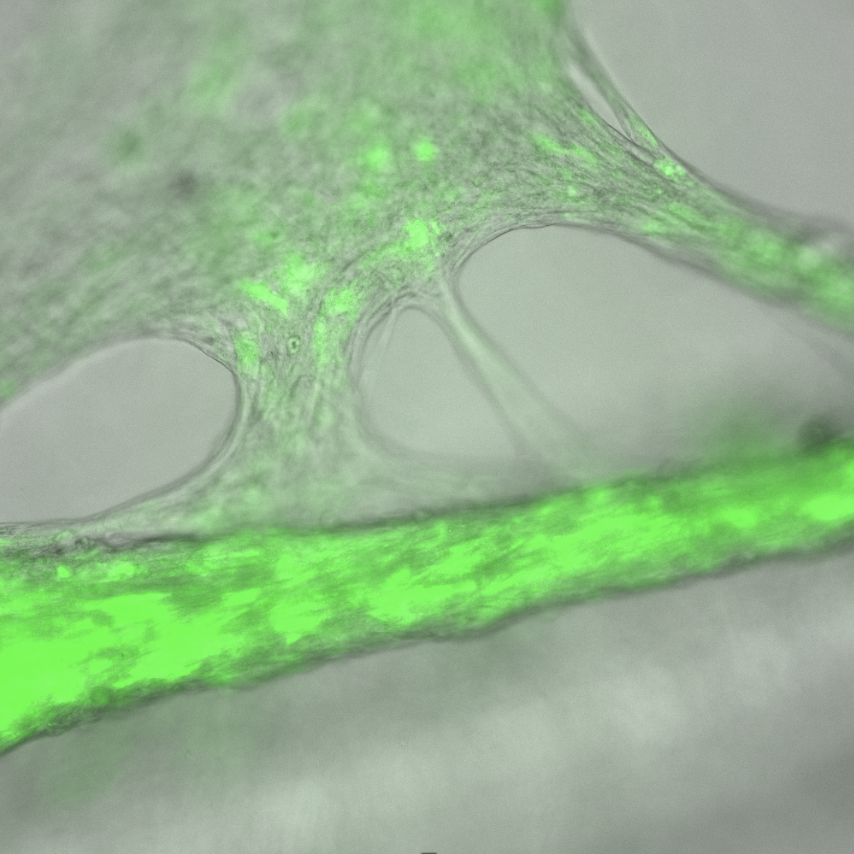
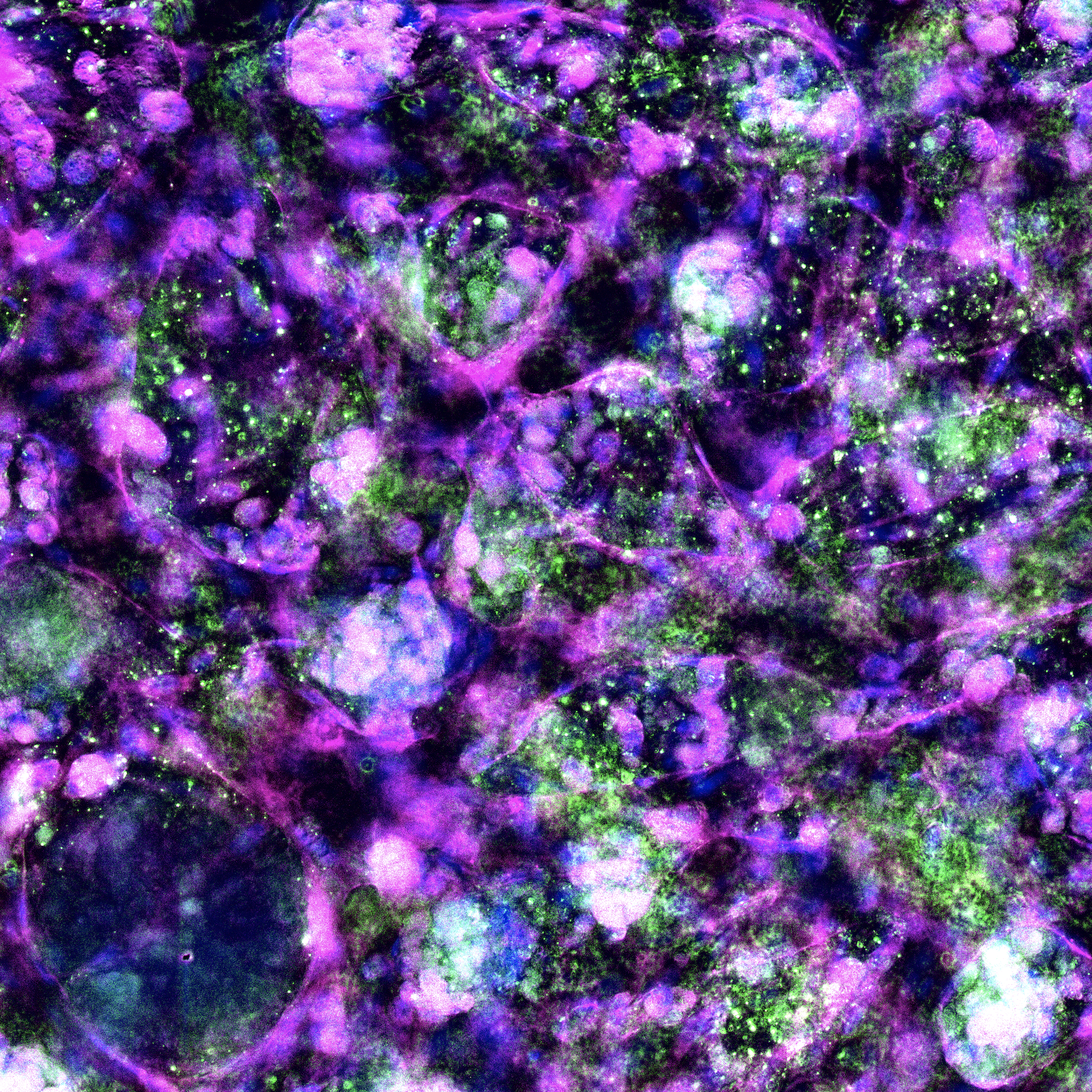
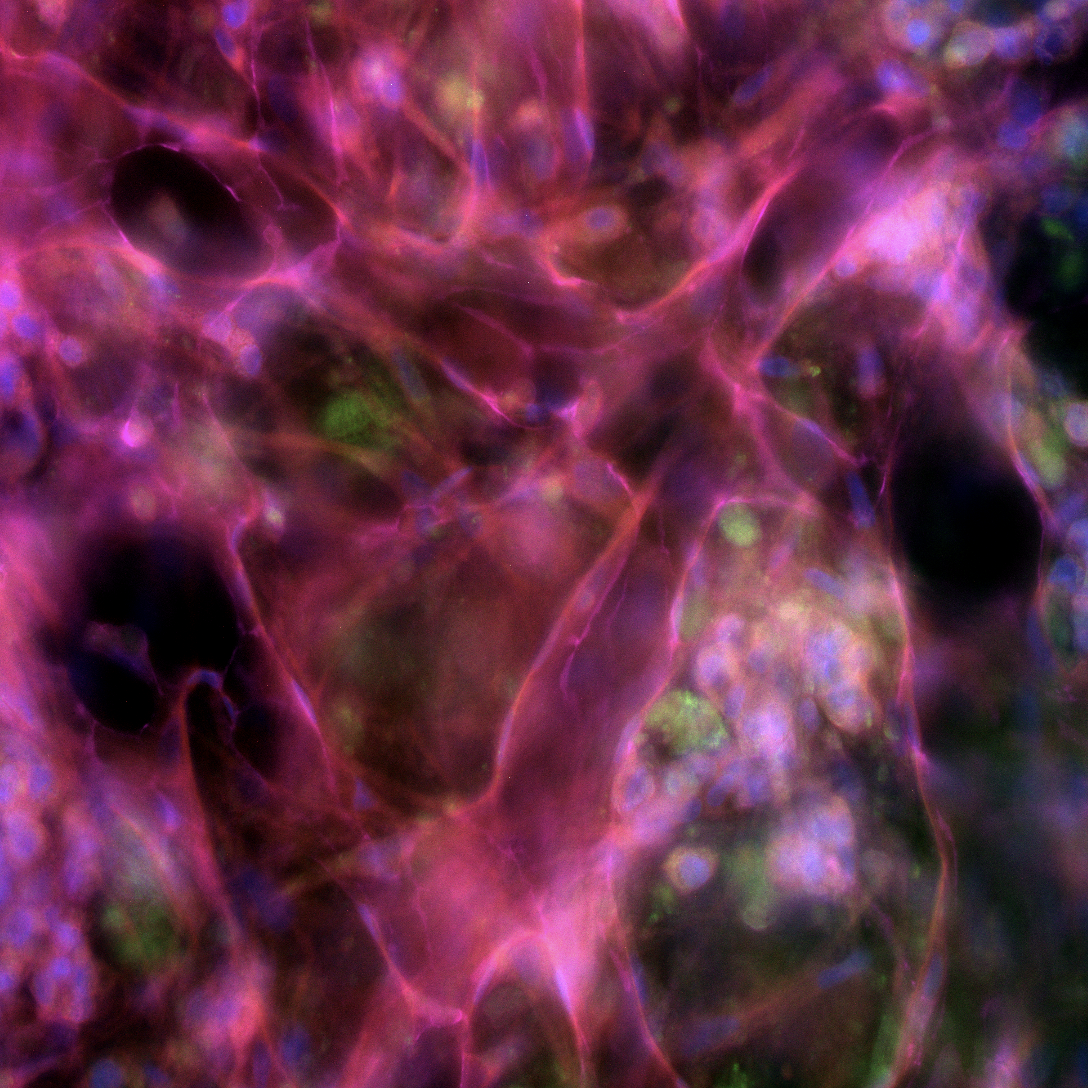
The missing vascular layer that makes organ models physiologically relevant
What: A proprietary platform integrating perfusable microvascular networks with 3D tissue constructs, enabling nutrient delivery, immune cell trafficking, and drug transport that mirror in vivo conditions.
Why: Most organ-on-a-chip models lack functional vasculature, limiting their ability to model immune-mediated injury, drug distribution, and long-term tissue viability.
Immunocompetent by design: Built on 15+ years of HLA immunobiology expertise. Supports donor-specific antibody exposure, immune cell trafficking, DC antigen presentation, T cell activation, immunosenescence modeling, and cytokine profiling.
Organ-specific models: Vascularized liver (regeneration, rejection, hepatotoxicity, embolization), blood-brain barrier (drug permeability, neurotoxicity), lymph node (cancer vaccine screening), glioblastoma (drug resistance, TME modeling).
Key publications: Adv. Mater. 2026 · Biofabrication 2025 · Lab Chip 2025




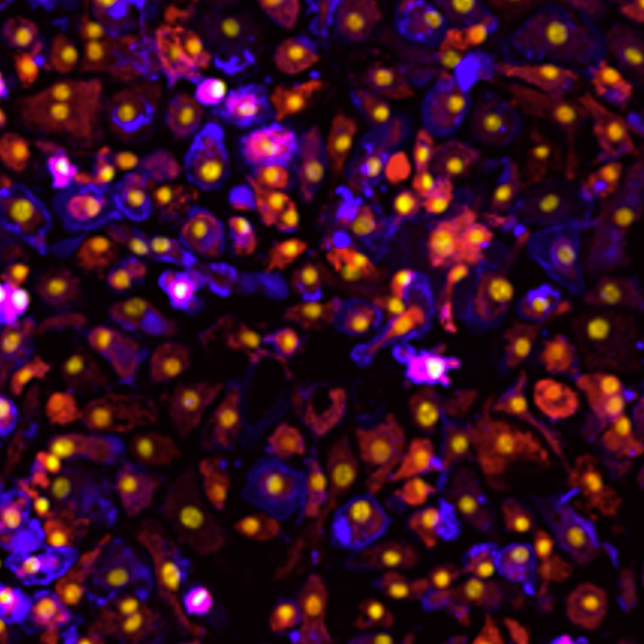
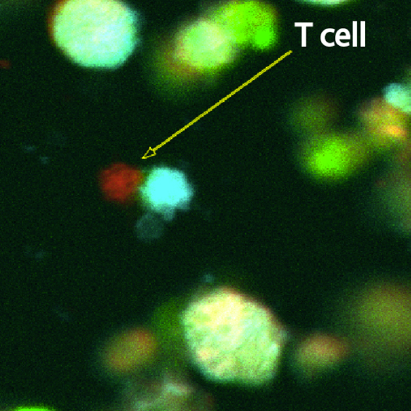
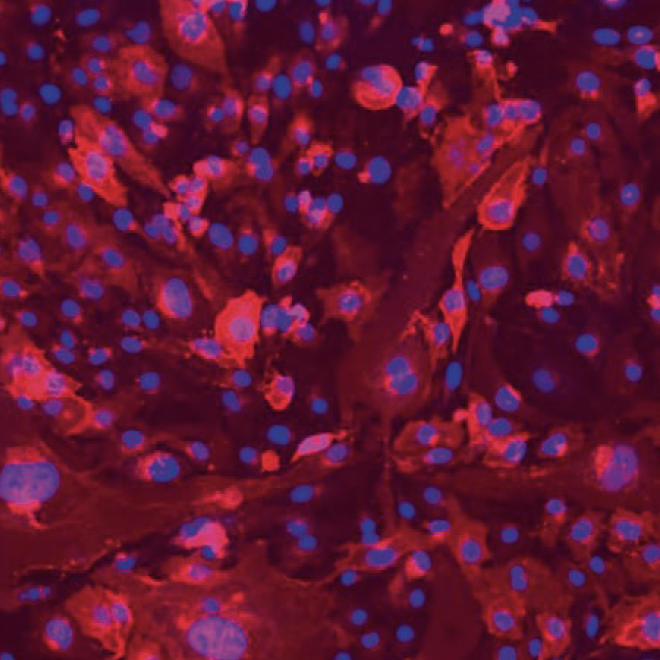

Accessible microfluidic infrastructure that brings organ models to any lab
What: Microfluidic chip systems that house and perfuse Tissueoid constructs with controlled flow, defined exposure conditions, and integrated electrode/sensor ports.
Why: Most OoC fabrication requires expensive cleanroom infrastructure. Our scalable thermoplastic approach makes the technology practical for any research setting.
Key features: Scalable thermoplastic fabrication, modular chip designs, integrated electrode and sensor ports, and perfusion systems supporting 28+ day culture.
Key publication: Small 2024


Transforming static tissue models into dynamically monitored living systems
What: Multiple sensing technologies embedded directly into OoC platforms for continuous, real-time monitoring without disrupting the biology.
Why: Traditional drug screening relies on endpoint assays that miss dynamic cellular responses, contributing to a >90% clinical trial failure rate.
PC-TIR optical biosensors: Label-free monitoring of antibody secretion and toxicity (with UT San Antonio).
TEER electrodes: Screen-printed electrodes for continuous barrier integrity measurement.
Electrochemical immunosensors: Reusable sensors for on-chip differentiation and biomarker secretion.
Contact lens biosensors: Microfluidic platforms for non-invasive tear exosome detection.
Key publications: Biosens. Bioelectron. 2025 · Small 2024



Advanced materials bridging tissue modeling and clinical therapeutics
What: Novel biomaterials enhancing OoC physiological relevance and enabling standalone clinical applications in wound healing and regenerative medicine.
Why: Faithful replication of native tissue properties is essential for both accurate modeling and direct therapeutic translation.
Hydrogel platforms: DES-based ionogels; peptide hydrogels; amyloid-mimicking hydrogels for neuronal studies.
Oxygen-generating biomaterials: Microparticles that downregulate HIF-1α and mitigate ischemic injury.
Natural polymer therapeutics: Latex-based dressings for wound healing, including a pilot clinical trial for diabetic foot ulcers.
Nanofibrous scaffolds: Granular porous microspheres for diabetic wound healing.
Key publications: Adv. Funct. Mater. 2023 · Acta Biomater. 2023



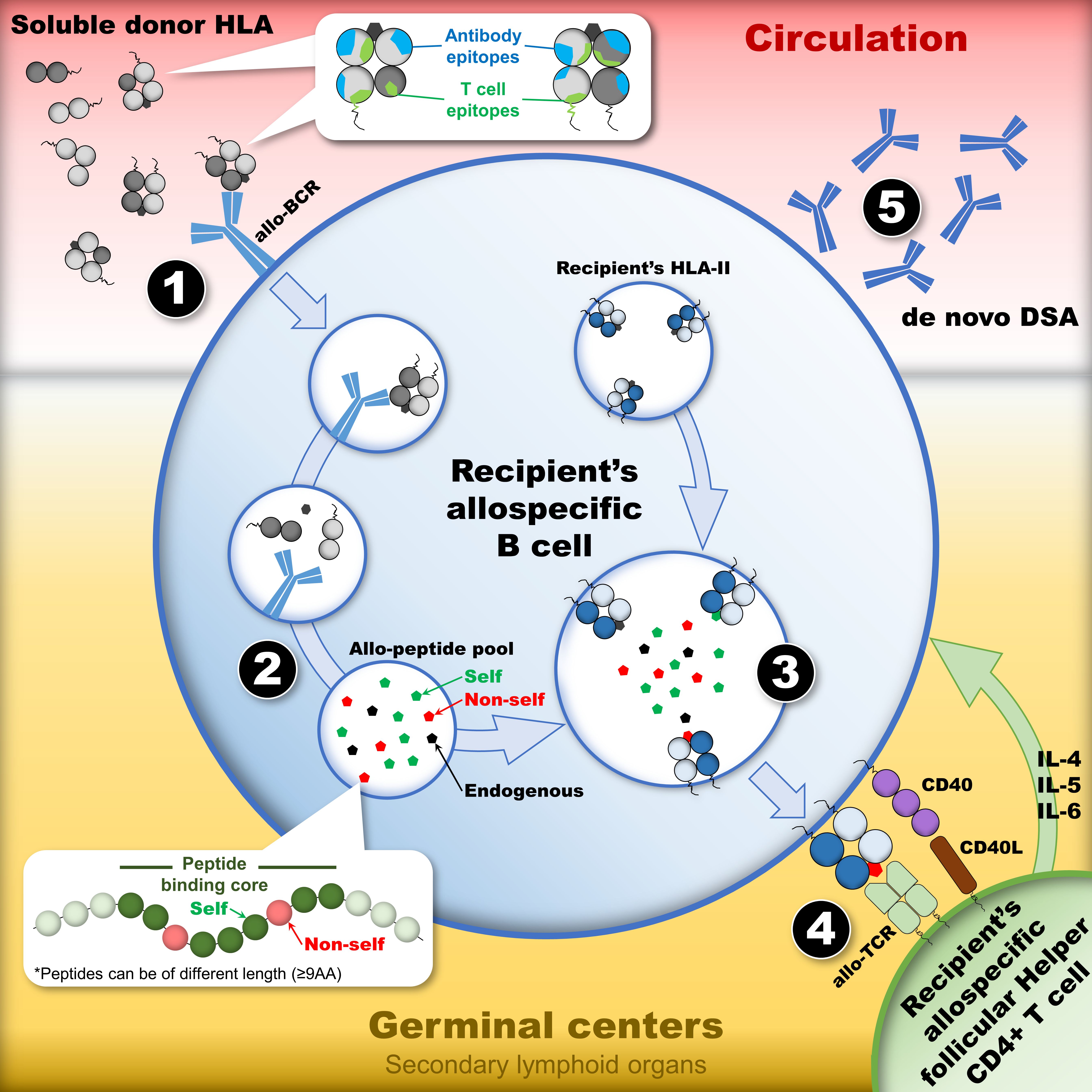
Comprehensive molecular characterization of HLA mismatches for transplant prediction
What: Feature engineering software generating 400+ molecular features from any donor-recipient HLA mismatch, identifying properties associated with antibody development and rejection.
Why: Existing tools assess HLA immunogenicity with single metrics. E3 integrates all approaches into one unified framework.
Feature coverage: HLAMatchmaker eplet scores, PIRCHE-II predicted epitopes, electrostatic/hydrophobicity differences, antibody-verified epitopes, plus novel VJLabs features.
Applications: Predicting de novo DSA development, identifying high-risk mismatches, optimizing donor-recipient matching, and supporting ML approaches to immunogenicity.
E3 has been used in multiple published studies and is being developed for broader availability.
Key publications: Antibodies 2024 · J Immunol Res 2017
Latest from VJLabs
 Grant
Grant VJLabs Awarded NIH R21 Grant for Xenotransplantation Research
VJLabs has been awarded an NIH R21 grant to advance xenotransplantation research using organ-on-a-chip technology.
Read More Publication
Publication New Vascularized Tissueoid-on-a-Chip for Liver Regeneration and Transplant Rejection
VJLabs published a milestone paper in Advanced Materials on our immunocompetent liver Tissueoid platform.
Read More Collaboration
Collaboration Strengthening Global Ties: Royal Belgian Economic Mission & Ghent University
The Terasaki Institute welcomed the Royal Belgian Economic Mission and Ghent University, strengthening international collaboration in biomedical innovation.
Read More Education
Education TIBI and CSUN Launch Partnership for Biomedical Research Education
The Terasaki Institute and Cal State Northridge launched a new partnership to advance biomedical research education and training.
Read More Publication
Publication 3D Microphysiological System Modeling Pericyte-Induced Chemoresistance in Glioblastoma
VJLabs developed a 3D microphysiological system that models pericyte-induced temozolomide resistance in glioblastoma.
Read More Publication
Publication Advancing Monoclonal Antibody Biomanufacturing
A new microfluidic-based biosensing platform for real-time monitoring of monoclonal antibody cellular secretion, developed with UT San Antonio.
Read More Publication
Publication Novel Liver Cancer-on-a-Chip Model for Embolic Agent Testing
A human vascularized liver cancer-on-a-chip model to evaluate vessel remodeling and cell death in response to embolic agents.
Read More Publication
Publication Cancer Vaccine Testing in Aging Populations
A new organ-on-a-chip platform recapitulating age-dependent immune responses for evaluating cancer vaccine performance in older adults.
Read More Grant
Grant $2.3 Million NIH Grant for Organ Transplantation Research
The Terasaki Institute was awarded a $2.3 million grant from NIH for organ transplantation research using organs-on-a-chip technology.
Read More Collaboration
Collaboration Mayo Clinic & Terasaki Institute Launch Collaboration for Transplant Innovation
Mayo Clinic and the Terasaki Institute launched a collaboration to advance transplant innovation through organ-on-a-chip technologies.
Read More Legacy
Legacy Remembering the Legacy of Dr. Paul I. Terasaki
Dr. Jucaud delivered a talk celebrating his mentor's life and moderated a Fireside Chat with leaders from UCLA, Cedars-Sinai, and One Lambda.
 Grant
Grant NIH Grant to Develop Allograft-Rejection-on-a-Chip Model
TIBI received an NIH grant to develop an allograft rejection-on-a-chip model for studying immune-mediated graft injury.
Read More Publication
Publication Screen-Printed Electrodes for Measuring Endothelial Barrier Integrity
Development of screen-printed electrodes for real-time TEER measurement of endothelial barrier integrity in organ-on-a-chip devices.
Read More Grant
Grant TIBI Awarded Grant from METAvivor
The Terasaki Institute received a grant from METAvivor to support metastatic breast cancer research.
Read MoreCurrent Members
An interdisciplinary team advancing organ-on-a-chip engineering, transplant immunology, and translational bioengineering.

Abdul R.C. Salih, Ph.D.

Arne Peirsman, M.D., Ph.D.

Danial Khorsandi, Ph.D.

Satoru Kawakita, M.S.

Bryan Hatherley, B.S.

Everardo Jauregui, B.S.
Alumni ▼
Click to expand - 27 former lab members
Research Output
52 original research papers, 25 reviews, 48 conference abstracts. Google Scholar →
2026 – 2025
- 2026 Salih ARC, Peirsman A, Khorsandi D, Ferrao R, Ferreira L, Kamaraj M, John JV, Baquerizo A, Jucaud V. Liver Tissueoid on-a-Chip Modeling Liver Regeneration and Allograft Rejection. Advanced Materials. e21178. doi:10.1002/adma.202521178
- 2025 Tang RC, et al. Rapid Point-of-Care Inflammatory Cytokine Monitoring during Normothermic Liver Perfusion via a Multiplexed Paper-Based Vertical Flow Assay. ACS Sensors. doi:10.1021/acssensors.5c01902
- 2025 Maity S, Hassani Najafabadi A, et al. Lymph node paracortex-inspired on-a-chip recapitulating immunosenescence: a cancer vaccine immunogenicity and antitumoral efficacy screening platform. Lab on a Chip. doi:10.1039/D5LC00533G
- 2025 Hernandez AL, et al. Continuous Optical Biosensing of IL-8 Cancer Biomarker Using a Multimodal Platform. Bioengineering. 12(10):1115. doi:10.3390/bioengineering12101115
- 2025 Khorsandi D, Yang JW, et al. Real-Time and Label-Free Monitoring of Monoclonal Antibody Secretion Rates Using a PC-TIR Biosensor. Biosensors and Bioelectronics. 117979. doi:10.1016/j.bios.2025.117979
Show all 52 original research papers ▼
2025
- 2025 Mohaghegh N, et al. Immunoengineering strategies using nanoparticles for obesity treatment. Nano Research. doi:10.26599/NR.2025.94907707
- 2025 Burd BS, et al. Can the insects Galleria mellonella and Tenebrio molitor be the future of plastic biodegradation? Science of The Total Environment. 969:178879. doi:10.1016/j.scitotenv.2025.178879
- 2024 Maity S, et al. Recent Developments in Glioblastoma-On-A-Chip for Advanced Drug Screening Applications. Small. e2405511. doi:10.1002/smll.202405511
- 2024 Khorsandi D, et al. Lab-on-a-chip: Unit Operations to Scale-up Strategies. In: Lab-on-a-chip Devices for Advanced Biomedicines. RSC. doi:10.1039/9781837673476
- 2024 Jucaud V. Allogeneic HLA Humoral Immunogenicity and the Prediction of Donor-Specific HLA Antibody Development. Antibodies. 13(3):61. doi:10.3390/antib13030061
Show all 25 review papers ▼
2025
- 2025 Salih ARC, Baquerizo A, Jucaud V. Vascularized Liver Tissueoid-on-a-Chip: An In Vitro Model for Studying Allograft Rejection. Am J Transplant. 25(8), S60. Oral presentation. World Transplant Congress 2025 (San Francisco).
- 2025 Jiang C, et al. Machine Learning-Based Risk Prediction of Allograft Rejection within One-Year Post-Transplant for Kidney Transplant Patients. Am J Transplant. 25(8), S67. Oral presentation. World Transplant Congress 2025 (San Francisco).
- 2025 Baquerizo A, Jucaud V, Keshavarz A, Markmann J. Xenogeneic Profiling of Human Plasma Protein Abundance Following Pig Hepatocyte Exposure during Bioartificial Liver Treatments. Am J Transplant. 25(8), S558. Poster presentation. World Transplant Congress 2025 (San Francisco).
- 2024 Hassani Najafabadi A, et al. Simvastatin-Loaded Polymeric Nanoparticles for Obesity Treatment. Oral. BMES 2024 (Baltimore).
- 2024 Nguyen HT, et al. Novel Vascularized Liver Tumor Model For Evaluation Of Embolic Agents. Oral. BMES 2024 (Baltimore).
Show all 48 conference abstracts ▼
We're Looking for Motivated Researchers
VJLabs welcomes inquiries from prospective postdoctoral fellows, graduate students, research associates, and summer interns passionate about organ-on-a-chip technologies, transplant immunology, biosensing, or biomaterials.
What We Offer
A collaborative, multidisciplinary research environment at the Terasaki Institute with access to state-of-the-art microfluidics, cell culture, and biosensing facilities. Mentorship, career development, and the opportunity to work on high-impact projects at the frontier of biomedical engineering.
How to Apply
Send your CV and a brief statement of research interests describing your background and how it aligns with VJLabs' mission.
Get in Touch
Dr. Vadim Jucaud
Assistant Professor
Terasaki Institute for Biomedical Innovation
Los Angeles, California
Email: vjucaud@terasaki.org




























