What we do - Our Platforms
Scientific advances have enabled us to know more about what makes each person unique down to the gene. It has enabled us to manipulate how a cell develops. It has provided us with mass amounts of data to study about the human condition. Here at the Terasaki Institute, we aim to utilize these advancements and develop them even further to deliver personalized diagnostics and solutions.
Our technology platforms allow us to study human disease and develop therapeutics on a personalized level. Physiological models can be personalized using organ-on-a-chip and advanced computational methods such as machine learning and deep learning. Therapeutics can be personalized using devices with sensors to deliver what is needed when it is needed. Damaged organs and tissues can be addressed with personalized transplants using biomaterials that are three-dimensionally printed.
Personalized Biomaterials

Biomaterials is a broad research area in our lab and our efforts lie at the interface of tissue engineering, microfluidics, and stem cell bioengineering. This includes synthesis and characterization of new biomaterials, the use of photo-crosslinkable biomaterials for patterning the surfaces, understanding cell-biomaterials interaction using high throughput screening and development of micro-/nanoparticles for controlled release. Currently, we have synthesized sugar-based photo-crosslinkable synthetic and natural polymers for tissue engineering applications. These polymers are being characterized for their mechanical properties, cytotoxicity, and ability to pattern themselves into various shapes and sizes. Natural and synthetic materials used in the lab include alginate, hyaluronic acid, collagen, polycaprolactone and polyglycerol sebacate for scaffold-based tissue.
Personalized Cells
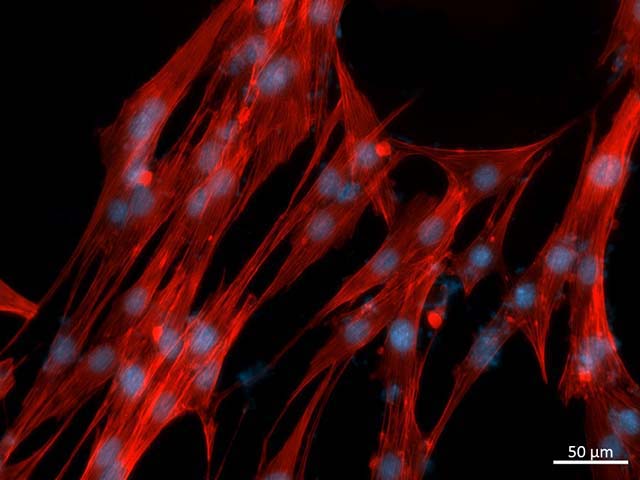
Immune engineering
The human immune system maintains the health of the body by blocking exogenous pathogens and removing endogenous transformed cells. Immunization against pathogens has been working for centuries by utilizing the immune system to maintain human health, and vaccination has been the standard medical method for decades. Recent breakthroughs in cancer immunotherapy, such as immune checkpoint discovery and chimeric antigen receptor (CAR) -T cell manipulation, have strengthened the oncology field. The Terasaki Institute offers another approach to tailoring precision medicine for phenotypic activation or immunotherapy where genetic engineers utilize the patient's immune system, including different types of immune cells. Training the patient's immune system to fight cancer is a multi-step biological process that is difficult to predict, particularly with regard to the effectiveness of treatment. The goal is to reprogram the patient's immune cells to directly recognize the signature antigens on the surface of the cancer cells using a synthetic biological approach. Through this strategy, the Terasaki Institute develops a faster and more controllable way to modulate the immune system.
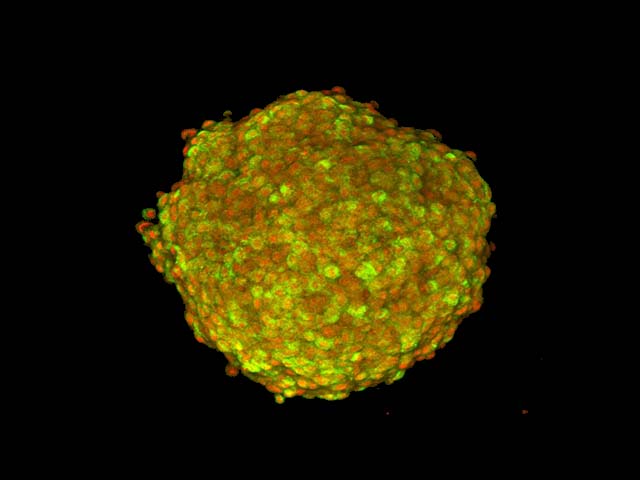
Stem Cell Engineering
Stem cells can be self-replicating and differentiate into the desired cell type. Various types of stem cells, including adipose derived stem cells, bone marrow derived stem cells, and induced pluripotent stem cells, can be used in regenerative medicine and produce cell replacement / regeneration therapies. The Terasaki Institute combines tissue engineering and genetic engineering techniques to develop stem cell-based treatment technologies, including MSC. In addition, The Terasaki Institute studies stem cell delivery technology with or without the help of a polymer scaffold. Our strategy is to develop a scaffold to provide a niche that mimics the mechanical properties of the natural extracellular matrix (ECM). Through this approach, our cell delivery scaffold can be integrated with signaling signals to regulate cell proliferation, differentiation and migration in a manner similar to the tissue engineering approach.
Personalized Device
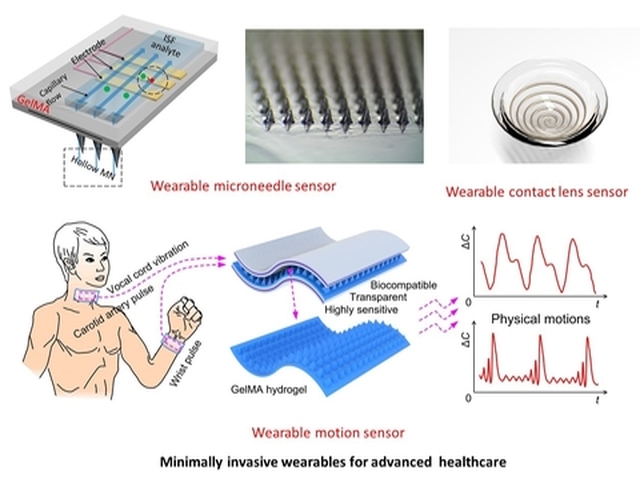
Sensors
There is a growing demand and interest in advanced biomedical devices that provide personalized healthcare. Most medical instruments are still accessible only in hospitals and research labs and can be performed only by professionals. Therefore, we need advanced biomedical devices that provide convenient and accessible monitoring of an individual’s health. Such devices can warn patients and caregivers at the earliest possible time and allow for appropriate medical intervention and care.
Wearable biosensors are devices aimed to present advanced tools for monitoring human health in realtime. These devices can detect motions of the human body, physiological signals on the skin, and biomarkers in bodily fluid. This health-relevant data will be collected with easy-to-wear devices and shared on the cloud.
Here at the Terasaki Institute, we are developing a multitude of smart wearable devices that can monitor human motion, map electrophysiological signals, and detect biomarkers of interest. In particular, we focus on building minimally invasive platforms such as microneedles and contact lenses, for wearable detection of health indicators in interstitial fluid and tears. We distinguish ourselves by using organic bioelectronic approaches. This includes the synthesis of soft conducting polymers and conductive hydrogels, the development of cutting-edge manufacturing technologies, and the fabrication of wearable organic bioelectronic sensors.
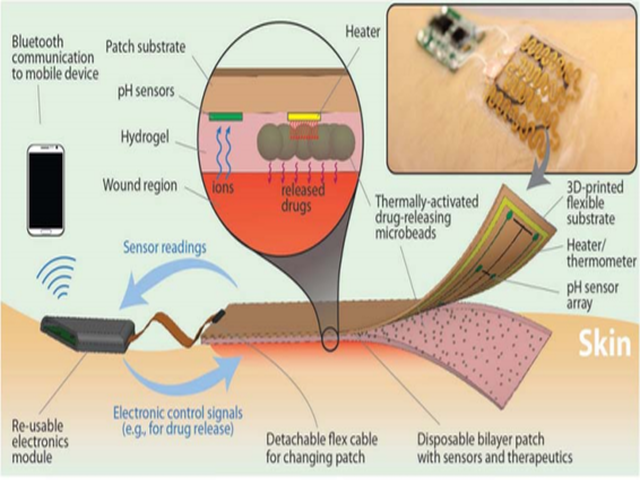
Theranostics
Theranostics, the combination of diagnostics and therapeutics, is the next step towards personalized medicine. Diagnostic devices that enable early detection provide useful and timely information about diseases that range from infection detection, disease diagnostics, etc. Furthermore, if one can combine early diagnostics with rapid therapy one can prevent numerous complications arising from late intervention. Along these lines, our group is working on a smart dressing for chronic wound healing applications. Chronic wounds are wounds that do not heal in a timely manner with a healing process exceeding 3-4 months. The smart dressing continuously monitors the wound area using pH and temperature sensors and in case of an infection delivers antibiotics for rapid intervention. By monitoring the wound in real-time, one can detect the infections instantaneously, submit the data to a medical professional and provide instant intervention preventing major medical complications such as severe infections, amputations, and extended hospital stays.
Over 6.5 million people in the USA suffer from burns and chronic wounds, and the imposed financial burden on the health care system is staggering. One of the major challenges in the healing process is bacterial infection, which not only impairs the wound’s healing process, but also exposes patients to other health risks due to the ineffectiveness of the skin’s physical barrier in the wound area. Detection of the infection process at an early stage and providing intervention in a rapid manner can prevent complications, including amputations. We have been developing a “smart wound dressing” platform that is able to continuously monitor the wound environment for bacterial infection using pH and temperature sensors and can provide local delivery of drugs for bacteria eradication. This platform is the first platform that demonstrates real-time sensing with a closed-loop feedback control for on-demand, localized delivery of drugs. The system consists of a multi-sensor chip that is connected to an electronics module used for acquisition, processing, and transmission of data.
Personalized Implants
Tissue engineering is an emerging field that aims to regenerate natural tissues and create new tissues using biological cells, biomaterials, biotechnology, and clinical medicine. The main goals of tissue engineering include providing the methods for replacement and regeneration of damaged tissues and creating tissue-like models for disease research. Recently, our team also combined scaffolds and cells to fabricate the cell-laden artificial tissues by utilizing 3D-printing techniques to create bone- and cardiovascular- like tissues. By utilizing these artificial tissues, we hope that it will allow scientists to deeply investigate the mechanisms behind the reparation of damaged tissues in regenerative medicine.
3D Printing
In biofabrication/3D bioprinting, the goal is to build artificial tissues that resemble the biological counterpart. However, tissues are wonderfully complex structures, so to engineer artificial versions of them that function properly, we have to recreate their complexity. Our tissue engineering team developed 3D bioprinting techniques to build 3D tissue constructs with viable cells, biomaterials, and bioactive molecules in a systematic and automated fashion. Specifically, our main focus lies on printing multi-bioink, cell-laden bioink, extracellular matrix (ECM)-based materials, or natural materials. These materials mimic complex tissue structure models in vitro such as liver, bone, or cardiovascular structures. When the technology is combined with patient-derived cells, it produces patient-specific tissue constructs, which can be used for personalized therapy and also as a means to personalize treatment. As a leader in tissue engineering, we continue to push the frontier of biofabrication/3D bioprinting to enable personalized medicine.
Personalized Nutrition

Personalized nutrition leverages human individuality to drive nutrition strategies that optimize health and prevent, manage and treat disease. Personalized nutrition is the concept of adapting food to individual needs. While it has become apparent that consumers respond differently to diet, depending on the content of food or nutritional supplements, it has been shown that having a generic formula that satisfies the needs of different individuals may not be working effectively per se. Personalization of dietary needs has emerged as a strategy which relies on the use of an individual’s information that takes into account a person’s genetic background, microbiome, geographical effectors and other parameters that affect the accessibility and metabolism of nutrients. This enables the delivery of highly-tailored, customizable nutritional advice and products.
In the Terasaki Institute for Biomedical Innovation, we are adapting our recent technologies for personalized medicine, tissue engineering and drug delivery to engineer personalized food, nutrition and related biosensing devices. Individualized 3D-printable nutritional snacks and the development and design of personalized protein and food resources, as well as nutritional delivery and sensing apparatuses are among the approaches that we are pursuing in our research.
Personalized Physiological Models
Organs-on-a-chip
The organs-on-a-chip platforms seek to recapitulate human organ functions at microscale by integrating microfluidic networks with three-dimensional tissue models, which are expected to provide robust and accurate predictions of drug/toxin effects in human bodies. Since induced pluripotent stem cells can be obtained from patient-derived cells such as skin or blood, it may enable personalized treatment or disease models with exactly the same genomic background as the patient. With recent advances in genome editing technology, cells with genetically desirable phenotypes, such as the expression of desired biomarkers, could be easily generated. Based on these high-end scientific endeavors, the Terasaki Institute is developing the most state-of-the-art organs-on-a-chip platforms with integrated bioreactors containing human tissue constructs and online sensors for real-time monitoring of both physical and biochemical parameters of the organoids. We are developing personalized physiological models with precisely designed genetic properties that provide a versatile platform to generate accurate medical therapies. Our efforts enable us to observe the basic function of cells and the input and output relationship allowing us to minimize the use of animal testing.

